History of the Law of the Conservation of Mass Antoine Lavoisier: A portrait of Antoine Lavoisier, the scientist credited with the discovery of the law of conservation of mass. Also, a molecule before it undergoes a chemical change. reactant: Any of the participants present at the start of a chemical reaction.product: A chemical substance formed as a result of a chemical reaction.law of conservation of mass: A law that states that mass cannot be created or destroyed it is merely rearranged.The law of conservation of mass is useful for a number of calculations and can be used to solve for unknown masses, such the amount of gas consumed or produced during a reaction.According to the law of conservation of mass, the mass of the products in a chemical reaction must equal the mass of the reactants.
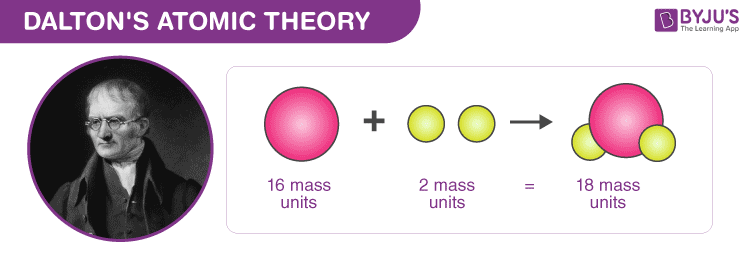
The law of conservation of mass states that mass in an isolated system is neither created nor destroyed by chemical reactions or physical transformations.However, the English chemist and meteorologist John Dalton is credited with the first modern atomic theory, as explained in his A New System of Chemical Philosophy. In 1661, Boyle presented a discussion of atoms in his The Sceptical Chymist. The concept of the atom was revisited and elaborated upon by many scientists and philosophers, including Galileo, Newton, Boyle, and Lavoisier. However, these ideas were largely ignored at the time, as most philosophers favored the Aristotelian perspective.
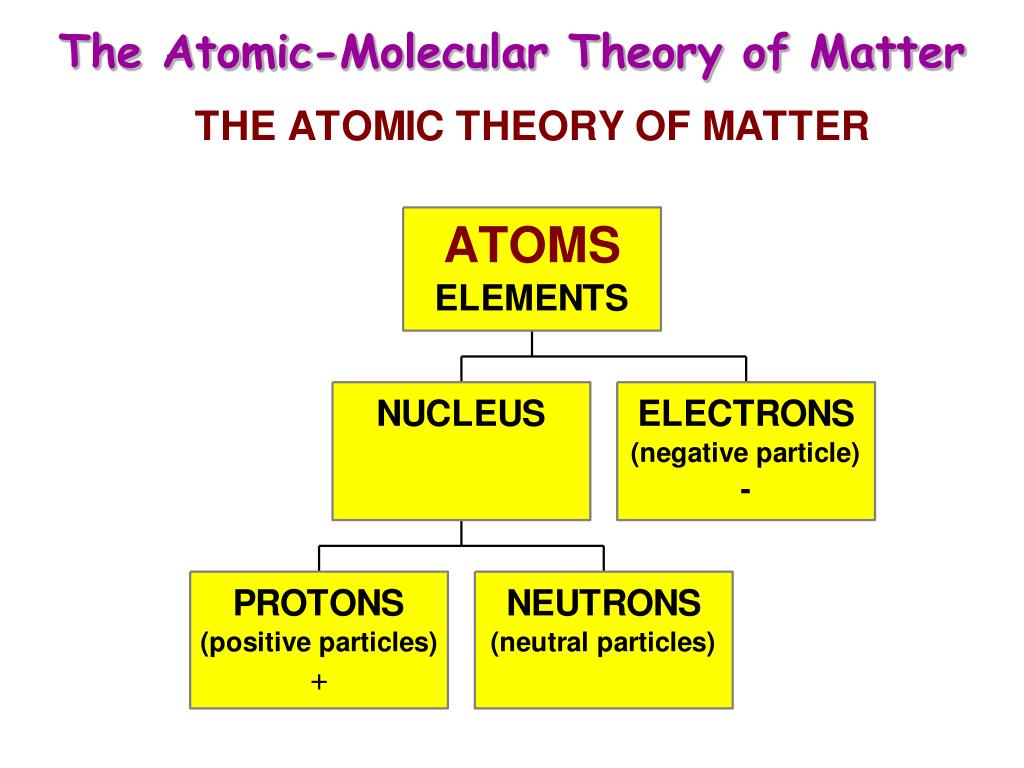
Democritus proposed that different types and combinations of these particles were responsible for the various forms of matter. The Greeks called these particles atomos, meaning indivisible, and the modern word “atom” is derived from this term. This idea was recorded as early as the fifth century BCE by Leucippus and Democritus. Matter is composed of indivisible building blocks.
